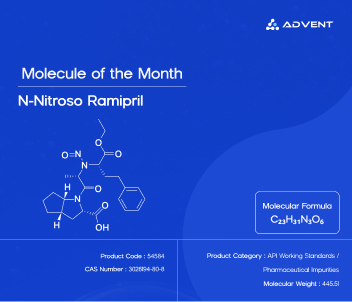
Ramipril is one of the most prescribed cardiovascular medications worldwide. An ACE inhibitor by mechanism, it has helped millions of patients manage hypertension and heart failure, improving quality of life and reducing the risk of serious cardiac events. But as with many established active pharmaceutical ingredients (APIs), the story does not end with the drug itself. Behind every manufactured molecule lies a complex chemistry — and sometimes, that chemistry gives rise to unwanted guests.
N-Nitroso Ramipril is one such guest. Chemically identified as (2S,3aS,6aS)-1-(N-((S)-1-ethoxy-1-oxo-4-phenylbutan-2-yl)-N-nitroso-L-alanyl)octahydrocyclopenta[b]pyrrole-2-carboxylic acid, this nitrosamine impurity has emerged as a subject of intense regulatory and scientific scrutiny. As Advent's Molecule of the Month for March 2026, it serves as a powerful reminder of why rigorous analytical science is inseparable from pharmaceutical safety.
What Is a Nitrosamine Impurity — and Why Does It Matter?
Nitrosamines are a class of chemical compounds that have gained significant regulatory attention over the past several years, following a series of high-profile recalls involving commonly used medications. Their concern stems from a well-established toxicological profile: many nitrosamines are classified as potential human carcinogens, capable of causing DNA damage at even trace concentrations.
In the context of Ramipril, N-Nitroso Ramipril can form during manufacturing processes or storage — particularly when secondary amines in the drug's structure react with nitrosating agents present in the environment or excipients. The window for its formation is small, but the implications are significant. Even at parts-per-million or parts-per-billion levels, the presence of this impurity must be identified, quantified, and controlled within acceptable intake limits established by global health authorities.
The Regulatory Imperative
International regulatory bodies — including the US FDA, the European Medicines Agency (EMA), and the International Council for Harmonisation (ICH) — have introduced stringent guidelines governing nitrosamine impurities in pharmaceutical products. ICH M7 and the FDA's nitrosamine guidance documents have defined acceptable daily intakes (ADIs) and mandated that manufacturers implement robust risk assessment and control strategies.
For drug developers and quality assurance teams, compliance is not merely a regulatory checkbox. It represents a commitment to patient safety. Detecting N-Nitroso Ramipril at trace levels requires validated analytical methods of exceptional sensitivity and specificity — typically leveraging LC-MS/MS (liquid chromatography coupled with tandem mass spectrometry) and other advanced techniques.
Advent's Role: Enabling Analytical Excellence
At Advent, our commitment is to empower pharmaceutical scientists and quality teams with the tools they need to do this work with confidence. N-Nitroso Ramipril is now available as a high-purity Working Standard — produced at milligram to gram scales with a purity exceeding 95% — precisely to support method development, method validation, and routine analytical testing in both domestic and international pharmaceutical settings.
Each lot of our N-Nitroso Ramipril Working Standard is accompanied by complete characterization data, including 1H NMR, Mass Spectrometry, Infrared Spectroscopy (IR), and Thermogravimetric Analysis (TGA). A Certificate of Analysis (CoA) with potency values is also provided, ensuring traceability and confidence across every stage of the analytical workflow.
The compound is available with ready stock and prompt delivery — recognizing that in pharmaceutical development timelines, speed of access to reference standards can be a critical factor.
Why This Molecule Deserves Your Attention
The presence of impurities like N-Nitroso Ramipril underscores a broader truth about modern pharmaceutical science: drug safety does not begin and end with the active molecule. It extends to every component, every process, and every potential degradation pathway. As the regulatory landscape continues to evolve, the demand for high-quality, well-characterized reference standards will only grow.
Whether you are in the early stages of developing an analytical method, conducting formal validation studies, or maintaining ongoing quality monitoring of a commercial product, having access to a reliable, high-purity standard is foundational. Advent's N-Nitroso Ramipril Working Standard is designed to meet that need — with the scientific rigor and supply reliability that modern pharmaceutical teams require.
Conclusion
N-Nitroso Ramipril represents more than a chemical entity — it reflects the evolving complexity of pharmaceutical quality assurance in a world where safety standards are rightly becoming more demanding. By highlighting this molecule in our March 2026 campaign, Advent reaffirms its mission: to be the trusted partner of pharmaceutical scientists navigating the frontier of impurity detection, characterization, and control.
As regulatory scrutiny intensifies and analytical capabilities advance, the industry needs reference standards it can depend on. Advent delivers exactly that.