The past decade has witnessed nothing short of a revolution in metabolic disease therapy. GLP-1 receptor agonists, led by Semaglutide, transformed the treatment landscape for type 2 diabetes and obesity, demonstrating that peptide-based drugs could deliver outcomes once thought impossible in chronic disease management. Building on that foundation, Tirzepatide — a dual GIP and GLP-1 receptor agonist — has emerged as a defining molecule of the next chapter, offering even greater glycaemic control and weight reduction in clinical outcomes.
Behind every therapeutic peptide, however, lies a chain of precisely engineered chemical steps. And within that chain, the integrity of each intermediate determines the quality and safety of the final drug substance. It is in this context that Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH takes centre stage as Advent's Molecule of the Month for April 2026.
Meet the Molecule: A Strategically Important Tetrapeptide
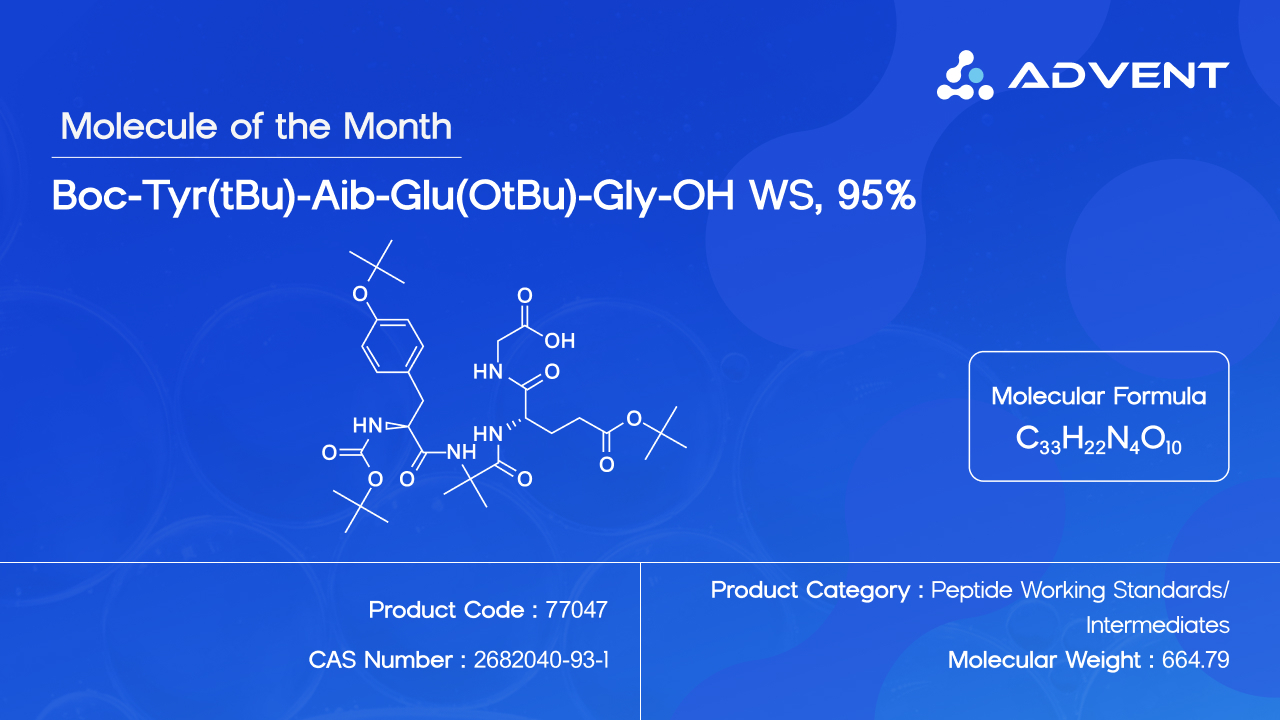
Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH (CAS No. 2682040-93-1; Product Code: 77047) is a protected tetrapeptide intermediate serving as the N-terminal building block of Tirzepatide. With a molecular weight of 664.79 g/mol and molecular formula CHNO, this compound encapsulates the complexity and precision that define modern peptide synthesis.
The molecule is composed of four amino acid residues, each carrying strategically placed protecting groups: a Boc (tert-butoxycarbonyl) group on the N-terminal tyrosine, tert-butyl (tBu) protection on the tyrosine hydroxyl and glutamic acid side chain, and a free C-terminal carboxylic acid on glycine. Together, these elements create a molecule that is chemically rich, synthetically demanding, and analytically significant.
The Role of Aib: A Conformational Game-Changer
One of the most scientifically compelling aspects of this tetrapeptide is the presence of Aib — alpha-aminoisobutyric acid — at the second position. Unlike standard proteinogenic amino acids, Aib is a non-coded, alpha,alpha-disubstituted residue that strongly induces helical conformations in peptide chains. Its inclusion in Tirzepatide's sequence is not incidental; it is a deliberate structural design choice that contributes to the compound's protease resistance and receptor binding profile.
For synthetic chemists, the incorporation of Aib presents unique challenges. Its steric bulk can reduce coupling efficiency during chain elongation, requiring optimised reagent systems and extended reaction times to achieve acceptable yields and purity. This is where process expertise becomes a true differentiator.
Synthesis at Advent: SPPS with Precision
At Advent, Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH has been successfully synthesised using Solid Phase Peptide Synthesis (SPPS) — the gold standard methodology for constructing protected peptide sequences with high fidelity. SPPS enables stepwise chain assembly with tight control over sequence integrity and impurity formation, which is critical when working with sterically hindered residues like Aib and orthogonally protected side chains.
The compound is currently developed at milligram to gram scales, with ongoing process optimisation focused on scale-up to meet growing commercial demand. This positions the tetrapeptide for dual application: as a synthetic intermediate in Tirzepatide API manufacturing, and as a peptide working standard for analytical purposes.
Analytical Significance: A Reference Material That Earns Its Place
Beyond its synthetic role, Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH carries substantial analytical value. In the development and validation of HPLC and LC-MS methods for Tirzepatide and its related substances, the availability of a well-characterised tetrapeptide intermediate is indispensable. It enables accurate impurity profiling, supports system suitability testing, and provides a reliable reference point for retention time and spectral matching.
As regulatory agencies increasingly scrutinise peptide API quality, having access to characterised intermediates for method validation and regulatory submissions is not a luxury — it is a necessity. Advent's working standard is accompanied by full characterisation data including 1H NMR, IR, Mass Spectrometry, and a Certificate of Analysis (CoA) with potency values, meeting the documentation standards expected by global regulatory bodies.
Handling and Storage: Getting the Details Right
Working with orthogonally protected peptides demands discipline beyond the laboratory bench. The stability of Boc and tBu protecting groups, while robust under neutral storage conditions, can be compromised by moisture, heat, and acidic or basic environments. To preserve sequence integrity and ensure reproducible analytical results, careful handling under controlled temperature and humidity conditions is essential.
Users working with this compound in HPLC and LC-MS workflows should be mindful of solvent selection and sample preparation protocols. Mobile phases and diluents must be chosen to maintain protecting group stability throughout the analysis, and column conditions should be optimised for the compound's unique physicochemical profile.
Looking Forward: The Tirzepatide Opportunity
The global Tirzepatide market is expanding rapidly. With approvals spanning multiple indications — including type 2 diabetes, obesity, and ongoing clinical investigations in cardiovascular disease — the demand for high-quality Tirzepatide intermediates and reference standards will only intensify. Pharmaceutical manufacturers, contract research organisations, and regulatory affairs teams all stand to benefit from reliable access to characterised building blocks like Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH.
At Advent, we are committed to staying ahead of this demand curve — investing in synthesis, characterisation, and scale-up to ensure our customers have what they need, when they need it.
Conclusion
Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH is more than a chemical intermediate. It is a window into the precision and complexity that underlies one of the most important peptide therapeutics of our era. By featuring this molecule in our April 2026 Molecule of the Month campaign, Advent reaffirms its position at the intersection of cutting-edge peptide science and pharmaceutical quality — enabling the next wave of therapeutic breakthroughs, one building block at a time.