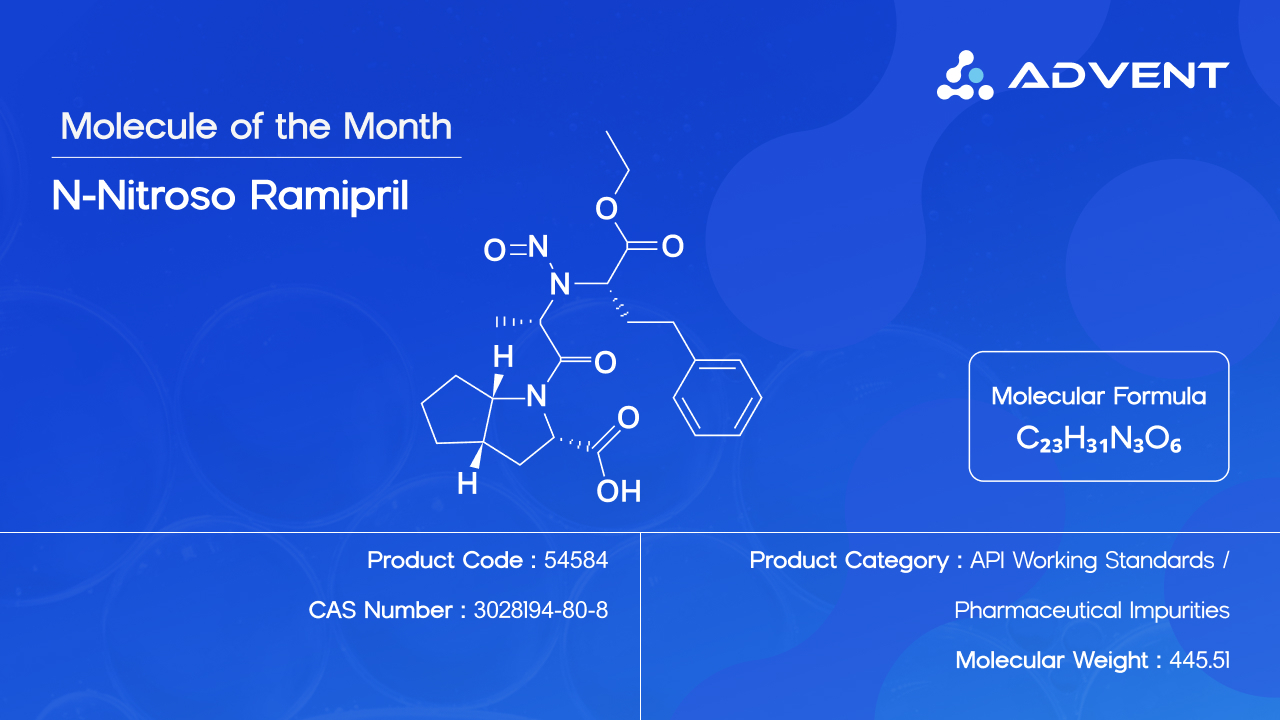
Advent, a leading provider of pharmaceutical reference standards and API working standards, today announced the availability of N-Nitroso Ramipril as a high-purity Working Standard for use in analytical method development, validation, and quality control testing. The product is offered with greater than 95% purity, ready stock availability, and full characterization documentation.
N-Nitroso Ramipril (CAS No. 3028194-80-8; Product Code: 54584) is a nitrosamine impurity associated with Ramipril, a widely used ACE inhibitor for the treatment of hypertension and heart failure. As a potential carcinogen under current international classification frameworks, its detection and control are subject to stringent regulatory requirements from the US FDA, EMA, and ICH guidance documents, including ICH M7.
Key Product Highlights:
The N-Nitroso Ramipril Working Standard is produced at milligram to gram scales and is accompanied by 1H NMR, Mass Spectrometry, IR, and TGA data, along with a Certificate of Analysis (CoA) with potency. It is available to pharmaceutical manufacturers in both domestic and overseas markets.
"Nitrosamine impurities represent one of the most pressing analytical challenges in pharmaceutical quality assurance today," said Dr. Rashmi Ranjan Mohanty, Director- Technical, Advent. "By making N-Nitroso Ramipril available as a well-characterized, high-purity working standard, we are helping our customers meet their regulatory obligations with confidence and precision."
The compound is available in the API Working Standards and Pharmaceutical Impurities category, with ready stock ensuring minimal lead times for time-sensitive projects. Pharmaceutical companies conducting risk assessments, method validation, or routine surveillance testing for nitrosamine impurities in Ramipril formulations will find this standard an essential addition to their reference material library.
For more information, to request a quotation, or to place an order, please contact Advent's technical sales team or visit our website.