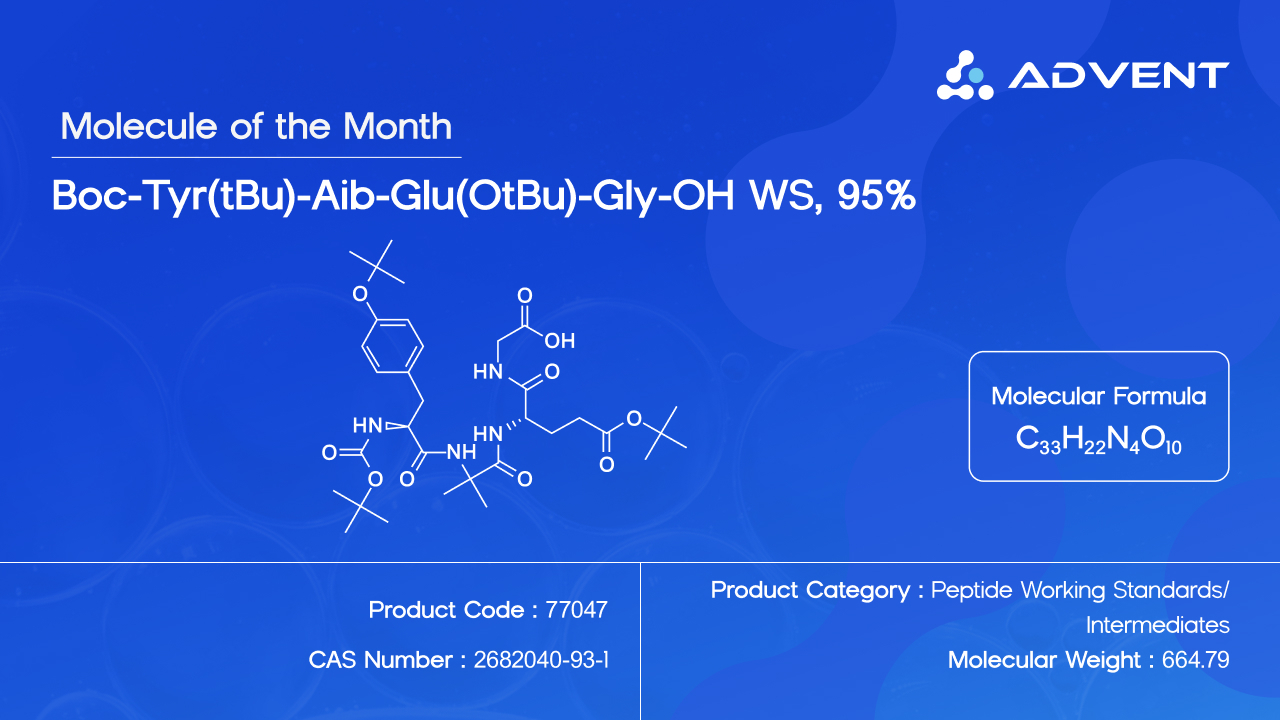
Advent, a trusted manufacturer and supplier of peptide working standards and pharmaceutical intermediates, today announced the availability of Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH (CAS No. 2682040-93-1; Product Code: 77047) as a high-purity Peptide Working Standard. The compound is produced using Solid Phase Peptide Synthesis (SPPS) at milligram to gram scales, with a purity of 95% or above, and is available for immediate dispatch to domestic and international customers.
This protected tetrapeptide serves as the N-terminal building block of Tirzepatide — the dual GIP/GLP-1 receptor agonist that has rapidly become one of the most significant peptide therapeutics in the treatment of type 2 diabetes and obesity. As global investment in Tirzepatide manufacturing and incretin-based therapies continues to grow, demand for well-characterised synthetic intermediates and reference standards for this molecule is rising in parallel.
Product Highlights:
Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH features orthogonally protected amino acids and the conformationally distinctive Aib residue. It is supplied with comprehensive characterisation data including 1H NMR, IR, and Mass Spectrometry, along with a Certificate of Analysis (CoA) providing potency values. The product is suitable for use in analytical method development, method validation, impurity profiling, and regulatory submissions related to Tirzepatide API manufacturing.
"As the global peptide therapeutics market matures, access to rigorously characterised intermediates becomes a cornerstone of both manufacturing quality and regulatory confidence," said Dr. Rashmi Ranjan Mohanty, Director- Technical, Advent. "Our Boc-Tyr(tBu)-Aib-Glu(OtBu)-Gly-OH Working Standard reflects our commitment to enabling peptide API development at every stage — from early research to commercial-scale production."
With ready stock and prompt delivery, Advent ensures that pharmaceutical development timelines are not constrained by reference material availability. The compound is listed under the Peptide Working Standards and Intermediates category and is accessible through Advent's global distribution network.
For further information, technical enquiries, or to request a quotation, please contact Advent's sales and support team.